Subscribe to RSS

DOI: 10.1055/s-0039-1693720
Effects of Neuroendocrine Centers on Germ and Germline Cells in Dendrobaena atheca Cernosvitov (Annelida: Lumbricidae)
Authors
Address for correspondence
Publication History
27 August 2018
20 May 2019
Publication Date:
08 August 2019 (online)
Abstract
The aim of the present comprehensive experimental study was to reveal the effects of the removal and the regeneration of the main neuroendocrine centers in the oogonia, oocytes and trophocytes in Dendrobaena atheca. Various types of serial sections of ovaries and cephalic regions were obtained. Four histological staining techniques were employed. In controlled preparations, the neurosecretory activities of A-cells and C-cells showed correlation with cellular activities of oogonia, oocytes and trophocytes. In experimental preparations, removal of A-cells caused abnormalities in oocytes and trophocytes. The regeneration of A-cells restored vitellogenesis and repaired abnormalities. In both preparations, C-cells remained aldehyde-fuchsin (AF)-positive. B-cells and U-cells remained AF-negative. The oogonia showed continuous mitotic divisions. Regenerated A-cells appeared in week 3, increased in number, but could not stop the abnormalities, as hormones were not transported due to the lack or scarcity of blood capillaries. Therefore, abnormalities increased extremely. Then, they stopped, decreased, and were repaired due to hormonal transport via fully reconstructed blood capillary plexuses. The main conclusions are: (1) oocytes and trophocytes are controlled by A-cells, since they exhibited prominent changes during the removal and regeneration of A-cells; (2) oogonia are controlled by C-cells, as they showed correlation of activities with C-cells and were not affected by the removal and regeneration of A-cells; (3) B-cells and U-cells remained inactive; hence, they have no role in oogonia divisions and vitellogenesis; and (4) delayed hormonal effects of regenerated A-cells were due to the lack or scarcity of blood capillaries. Therefore, fully reconstructed blood capillary beds in the A-cells area are indispensable for hormonal diffusion, transport and effectivity.
Keywords
main neuroendocrine centers - C-cells - fully reconstructed blood capillary plexuses - germ cells - germline cellsIntroduction
Sexual reproduction in annelids is chiefly controlled by neurosecretory cells (NSCs) of the main neuroendocrine centers (MNECs).[1] [2] [3] [4] [5] [6] [7] [8] [9] [10] The MNECs in earthworms consist of suprapharyngeal nerve ganglia (SPPNG) and subpharyngeal nerve ganglia (SBPNG). Neurosecretory cells are heavily concentrated in these two large paired complex ganglia, but are not only restricted to these two big centers due to the phenomenon of redundancy. The segmental nerve ganglia (SNG), especially the anterior ones, do contain two important types of active NSCs.[11] [12] [13] Aldehyde-fuchsin (AF)-positive NSCs have also been discovered in ganglia of the vegetative nervous system (VNS)[14] in earthworms. So, the removal of the whole MNECs in earthworms does not mean at all that all NSCs have been removed.
The role of neuroendocrine centers in the reproduction of Hirudinea and Polychaeta has been extensively studied.[1] [3] [4] [5] [6] [15] [16] [17] Conversely, very few experimental studies have been done in this field in Oligochaeta.[2] [7] [8] [9] [10] [18] Additionally, these studies were restricted to certain parts of the MNECs or to certain types of germ and germline cells of the ovary. Also, the B-cells and C-cells of the SNG, and the B-cells, U-cells and C-cells of the SBPNG were entirely neglected and ignored.
It is noteworthy to mention that the NSCs of the MNECs of Dendrobaena atheca have previously been thoroughly investigated.[11] [12] [13] [14] Two types of NSCs, A-cells and B-cells, were described in the SPPNG (brain, cerebral ganglia), whereas three types of NSCs, B-cells, C-cells and U-cells of Hubl, were reported in the SBPNG. The SNG, particularly the anterior ones, exhibited two types of NSCs, B-cells and C-cells. Also, the ovaries of the same earthworm have recently been histomorphologically studied in detail.[9] [10] [19] Three types of germ and germline cells, the oogonia, oocytes and trophocytes, were described, in addition to three types of somatic cells.
The present experimental study, therefore, was undertaken due to: (1) the scarcity of comprehensive experimental studies, which show correlation between the activities of the various types of NSCs in the MNECs and of the various types of germ and germline cells in the ovaries of earthworms. Such studies, in fact, are very rare, if any at all exists; (2) the previous experimental studies have shown the effect of the removal and regeneration of only one type of NSCs, the A-cells, of the SPPNG. They have entirely neglected three very important types of NSCs, B-cells, C-cells and U-cells of the SBPNG and of the SNG; (3) the total monitoring period of the experiment in previous studies was relatively short (∼3 weeks). A longer monitoring period is certainly more preferable and may lead to better results and conclusions. Therefore, the present experimental study chiefly aimed to: (1) search for a correlation between neurosecretory activities of various types of NSCs in the MNECs and the cellular activities of various types of germ and germline cells in the ovaries of controlled D. atheca during the breeding season (autumn); (2) to reveal the effects of the removal and regeneration of various types of NSCs of the MNECs (SPPNG and SBPNG), in addition to the SNG, on the oogonia, oocytes and trophocytes in experimental earthworms; and (3) to monitor the control and experimental earthworms comparatively for a longer time (8 weeks), aiming to obtain explicit results and conclusions.
Materials and Methods
About 300 sexually mature earthworms, D. atheca Cernosvitov, were collected during the breeding season (autumn) from the Khanke Community of the Duhok Governorate, Kurdistan Regional Government of Iraq. They were kept as stock in 2 large plastic containers (50 cm in length, 35 cm in width, and 25 cm in depth) in moist soil brought from the place of collection. They were reared for 2 weeks in the laboratory for acclimatization under a temperature ranging from 20 to 24°C, and under a relative humidity ranging from 55 to 60%, before starting the true experimental work. An extra separate quantity of moist soil was brought from the region of collection to the laboratory. It was used when needed.
Acclimatized earthworms were divided into three groups: experimental, control, and stock. The experimental group included 64 specimens, which were divided into 4 replicates, each containing 16 worms that were reared in 4 small plastic containers (20 cm in length, 14 cm in width, and 5 cm in depth). To ensure the removal of the whole MNECs (SPPNG and SBPNG), the 1st 6 anterior segments, in addition to the prostomium, were amputated from each worm. The extirpated cephalic segments were eliminated, whereas the decapitated experimental worms were returned back to their small containers. They were monitored for 8 weeks to detect the effects of the removal and regeneration of various types of NSCs of the MNECs on various types of germ and germline cells (oogonia, oocytes and trophocytes) of the ovaries. The control group was also composed of 64 specimens, which were divided into 4 replicates, each containing 16 specimens that were reared in small plastic containers. The remaining earthworms were reared as a stock or as a spare group in two large containers. These worms were used when needed. All of the containers were almost filled with moist soil and covered by gauze. The moist soil of all containers was replaced twice at equal intervals.
At the time of the decapitation of the experimental earthworms, eight control earthworms, two from each replicate, were randomly chosen. The 1st 6 anterior segments containing the MNECs (SPPNG and SBPNG), in addition to segments XI to XV, containing the ovaries, were excised from each worm and were fixed in alcoholic Bouin fluid (Duboscq-Brasil fixative). They were used to reveal the secretory activity of various types of NSCs in MNECs and the cellular activity of the various types of germ and germline cells in the ovaries.
At the end of week 1 of the experiment, that is, 1 week after the decapitation of the experimental worms, 8 decapitated experimental worms, 2 from each replicate, were randomly chosen. Four anterior segments were cut from each of them and were fixed in the same fixative. These were used to follow the regeneration of the NSCs of the MNECs. Additionally, the segments XI to XV of the same worms were also cut and fixed to detect the effect of the removal and regeneration of the whole MNECs on the oogonia, oocytes and trophocytes in the ovaries. At the same time, eight control worms, two from each replicate, were also randomly chosen and used. The 1st 6 anterior segments of each of these worms were extirpated and fixed to follow the neurosecretory activities of all types of the NSCs in the MNECs. Additionally, the segments XI to XV of the same worms were also excised and fixed to detect the activities of germ and germline cells in the ovaries in the presence of the NSCs of the MNECs. This act was repeated at the end of week 2 up to week 8.
Four textbooks[20] [21] [22] [23] of microtechniques were used for various histological techniques. The fixed materials of the MNECs and the ovaries of both control and experimental worms were subjected to further histological procedures, such as the removal of the excess fixative, dehydration, clearing, wax infiltration, blocks preparation, and trimming. Serial cross, longitudinal and horizontal sections of a thickness between 3 and 9 µm were obtained, using the rotary microtome (KEDEE, CHINA). The histological staining techniques used were: (1) Gill III hematoxylin-alcoholic eosin (G3HE) (2) Heidenhain azan modification of Mallory triple stain (HA) (3) the chrome-alum hematoxylin phloxine (CHP) technique, and (4) the AF technique. The CHP and AF techniques are usually used to detect NSCs, neurosecretory material (NSM), and neurosecretory granules, which are stained purple and black to black blue with the AF and CHP techniques, respectively. The stained preparations of ovaries and MNECs were thoroughly examined under the compound light microscope (Amscope). Colored photomicrographs of the stained sections of ovaries and MNECs were obtained using a digital camera (Samsung 16 mega pixels).
Results and Discussion
Neurosecretory Cells of the Main Neuroendocrine Centers and Germ and Germline Cells of Ovaries in the Control Group
The control histological preparations of the MNECs (SPPNG and SBPNG) and the ovaries of the earthworms were thoroughly examined. A-cells ([Figs. 1A-C]) of the SPPNG and C-cells ([Figs. 1D] and [E]) of the SBPNG, in addition to the SNG, showed remarkably strong neurosecretory activity. They showed very strong staining affinity for both AF and CHP techniques. Neurosecretions were detected in both perikarya and axons ([Fig. 1C]) of these cells. The active NSCs and the NSM were stained purple and black to black blue with AF and CHP, respectively. Contrariwise, the NSCs of type B ([Figs. 1A-D]), in the SPPNG, SBPNG, and SNG showed no NSM. They appeared inactive and remained AF- and CHP-negative in control and experimental worms throughout the tenure of the experiment. These results are in line with those reported by Hagadorn,[4] by Kulkarni et al,[6] and by Khalid.[9] They have shown that A-cells and not B-cells of the SPPNG or of the supraesophageal ganglia exhibited strong secretory activities during the reproductive cycle. All annelids endocrinologists agree that the cerebral ganglia or the SPPNG have an indispensable role in the regulation of the reproductive cycle due to the presence of a huge number of strongly AF- and CHP-positive type-A NSCs, which are inevitable for normal vitellogenesis and egg production. The U-cells ([Fig. 1D]) of the SBPNG, like the B-cells, were inactive and showed no NSM throughout the tenure of the experiment. This clearly indicates that both cell types play no role in oogonia division or in vitellogenesis.
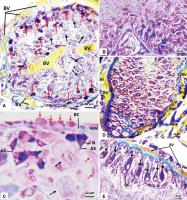

The oogonia, oocytes and trophocytes in the ovaries exhibited normal morphology, growth, division, and vitellogenesis. The oogonia ([Figs. 2A] and [B]) showed normal morphological features, continuous active mitotic divisions, and the peripherally ornamented nuclear membrane with chromatin granules, a distinct characteristic of the nuclei of oogonia. The oocytes ([Fig. 2C]) also showed normal morphological characteristics, normal growth, signs of active vitellogenesis with deposition of two zones of yolk granules, central and peripheral. They were also devoid of vacuoles and exhibited a quite clear, smooth, uniform, and mostly regular border with zona pellucida (ZP). The trophocytes, ([Figs. 2D] and [E]) appeared in groups or chains accompanying the oocytes. They exhibited normal outlines, strongly stained cytoplasm, nuclear membrane, nucleolus, and chromatin network. These results are very similar to those reported in Dendrobaena veneta.[8]


The controlled results of the NSCs of the MNECs and of the germ and germline cells of the ovaries were carefully examined. Positive coincident correlations between the activities of the cells of the MNECs and the cells of the ovaries were detected. The strong neurosecretory activities of the A-cells and C-cells of the MNECs coincided with active vitellogenesis and mitotic divisions in the oocytes, trophocytes and oogonia. This simultaneous correlation in activities of these cells strongly suggests that A-cells and C-cells are the source of some neurosecretions (gonadotropins), which help regulate the cellular activities in oogonia, oocytes and trophocytes. Conversely, both B-cells and U-cells remained inert throughout the tenure of the experiment in the control and experimental groups, thereby indicating that they play no role in oogonia divisions and vitellogenesis. Actually, these facts were further clarified, supported and confirmed by the results of the experimental section of the present study.
Neurosecretory Cells of the Main Neuroendocrine Centers and Germ and Germline Cells of Ovaries in the Experimental Group
The experimental histological preparations were also carefully examined. To understand the results of the experimental part easily and precisely, they were divided into three sections, based both on the activity and on the effectivity of the NSCs of the regenerated MNECs or of the SPPNG.
Section One
Section one included the results of 2 weeks, week 1 (W1), and week 2 (W2). No stainable or active NSCs were detected in the regenerated MNECs in these 2 weeks. They were not yet differentiated or fully formed. Furthermore, the active NSCs of the original MNECs were entirely amputated or removed from all experimental worms at the beginning of W1. Hence, both the activity and the effectivity of the NSCs of the regenerated MNECs were nil, since in W1 and W2, AF- and/or CHP-positive NSCs were entirely absent. The germ and germline cells, therefore, were completely deprived of neurosecretions or of gonadotropins of both the regenerated and original MNECs.
The gonadotropin-deprived oocytes ([Fig. 3A]) in W1 showed several distinct abnormal changes, such as few peripheral vacuoles, irregular border of plasma membrane, little distortion, degeneration, and withdrawal of ooplasm from the ZP. The trophocytes, contrariwise, showed no detectable abnormal changes in W1. At the end of W2, signs of disintegration, distortion, shrinkage, and vacuolization of ooplasm clearly increased ([Fig. 3B]). Vacuoles inside the ooplasm increased in number, size, and dispersion ([Fig. 3B]). Trophocytes ([Fig. 3C]) in W2 showed some signs of degeneration and distortion in the peripheries. Conversely, the oogonia were not affected at all by the removal of NSCs of the MNECs and showed no abnormal changes. They remained normal, active, and continued mitotic divisions as those of the control animals ([Figs. 2A] and [B]) in both weeks. These results are, in fact, very similar to those reported in D. veneta.[8] It was also shown that the oogonia remained normal and continued mitotic divisions, although they were deprived of gonadotropin (neurosecretion). Hence, it was assumed[8] that the oogonia were not controlled by neurosecretions, since they were able to divide in absence of active NSCs of the MNECs.


Although the results of these two studies appear to be very similar, the evidence provided and the conclusions deduced in each study are, in fact, quite different. It is noteworthy to mention that the removal of the whole MNECs (SPPNG and SBPNG) in earthworms does not mean at all that all NSCs have been removed. Undoubtedly, NSCs are extremely concentrated in the SPPNG and in the SBPNG, but are not only restricted to these two large paired complex ganglia. In the presence and/or in the absence of the MNECs, two types of NSCs, B-cells and C-cells, do exist in almost all SNG. They do form very important groups of active NSCs, especially in the anterior SNG.[11] [12] [13] This unique characteristic is due to a phenomenon called redundancy in these metameric invertebrates, that is, some structures or organs, such as the nerve ganglia, nephridia, blood vessels, setae, etc., are repeated (with few exceptions) in each body segment. Therefore, one should be quite aware when searching the neurosecretory system in these segmented worms, and should not neglect or ignore at all the role of B-cells and C-cells of the SNG. These cells should be seriously considered in any neuroendocrinological investigation in earthworms.
The results of the present study strongly suggest that oogonia are controlled by neurosecretion of the C-cells of the SNG and of the SBPNG (when present). This suggestion is supported by three strong evidence. First, oogonia were not affected at all by the removal and/or regeneration of the A-cells or the MNECs. So, this clearly indicates that they are not controlled by A-cells. Second, in W1 and W2, C-cells were the only type of active NSCs present in the anterior SNG. The regenerated NSCs were not yet formed, and the original ones were amputated. Third, due to the coincident correlation between the active secretory state of the C-cells and the active mitotic divisions of the oogonia, not only during these 2 weeks, but throughout the tenure of the experiment in both the control ([Figs. 1D] and [E]) and experimental ([Figs. 3D] and [E]) groups. This concurrent correlation in the secretory activity of the C-cells and the active oogonia proliferation, especially in absence of active A-cells (W1 and W2), clearly indicates and strongly suggests that the oogonia are controlled by neurosecretion of the C-cells of the SNG and the SBPNG.
Section Two
Section two included the results of 3 weeks, week 3 (W3), week 4 (W4), and week 5 (W5). The regenerated MNECs were in advanced stages of regeneration at the end of W3. They clearly showed fully formed and very active regenerated NSCs. In fact, they were few in number, but exhibited very strong AF- and CHP-positivity. A-cells were seen in the SPPNG, and C-cells in the SBPNG and in the SNG. Although the regenerated A-cells ([Fig. 3F]) of the MNECs in W3 (as well as in W4 and W5) appeared morphologically very active, nevertheless they remained functionally ineffective, since they were unable to exert any positive hormonal effect. Therefore, A-cells in W3, W4 and W5 showed very strong activity, but no effectivity at all. Hormonal failure was, certainly, not due to low hormonal concentration or titer, since the active A-cells remained helpless even after they significantly increased in number and in secretory activity in W4 and W5. Even then, they could neither stop nor decrease the abnormal changes in oocytes and trophocytes. Therefore, all deteriorations, such as degeneration, destruction, disintegration, distortion, vacuolization, and withdrawal of the ooplasm, increased extremely ([Fig. 4A]) and reached their peak in W5. The trophocytes also showed enormous signs of degeneration ([Fig. 4A]) in W5. Vacuoles increased extremely in number, size and spreading, and almost filled the ooplasm, thereby the oocytes showed a sieve-like appearance ([Fig. 4A]). In fact, some oocytes and trophocytes, at the end of W5, were severely damaged and almost entirely destroyed ([Fig. 4A]). Destruction of the oocytes and of the trophocytes due to the lack of neurosecretion of active A-cell of the SPPNG certainly means that no eggs will be produced. Therefore, the role of the SPPNG in reproduction is indispensable. Conversely, the oogonia remained normal, active, continued proliferations, and also exhibited a coincident correlation the activity of the C-cells in these 3 weeks. Since the abnormalities increased extremely from W3 to W5, the results of the present study disagree with those reported by Siekierska[8] in D. veneta. In D. veneta, it was shown that the degenerative changes decreased in oocytes and trophocytes 20 days (∼ 3 weeks; short monitoring period) after decerebration.


On the other hand keen observation and careful examination of the regenerated MNECs, the active A-cells and the area of A-cells revealed the real cause of the delay of positive hormonal effects. It was shown that the area of active A-cells was entirely devoid of blood capillaries ([Fig. 3F]) in W3 and W4. No blood capillaries were seen at all in the regenerated area of A-cells. They were certainly seen outside the sheath of the SPPNG. However, in W5, the A-cells area was encroached upon by very few blood capillaries ([Fig. 4B]), which appeared, due to their scarcity, to be inadequate for the diffusion and transport of A-cells neurosecretion to ovaries. These results of the present study are in line with those reported in Allolobohora terrestris.[2] It was reported that, 3 weeks after the removal of the MNECs or of the SPPNG, it was regenerated in an advanced stages (with active NSCs), but it was still ineffective. In fact, the NSCs were very active, but remained ineffective, since the neurosecretion (neurohormone) was not transported to target cells due to the lack of fully reconstructed blood capillary networks in the area of active regenerated NSCs. Thus, deteriorations in the present study reached the maximum due to the lack of competent blood capillary beds. Therefore, a fully reconstructed blood capillary plexus is indispensable for hormonal diffusion, transport and effectivity.
It is well-known that all endocrine glands or systems, whether neural or non-neural, in invertebrates or vertebrates, are ductless glands. So, they do need a medium (blood, body fluid), which conveys their secretions to target cells. Therefore, they are always richly supplied with blood capillaries forming the so-called “elementary neurohemal structures or organs or storage zones”,[11] [12] [13] which nourish the NSCs and assist diffusion and transport of the released hormones. A large blood vessel, usually abutting the posterior part ([Fig. 1B]) of the SPPNG, does participate in the formation of these blood networks in the area of A-cells and in the periganglionic capsule (PGC).[11] [12] [13]
Section Three
Section three included the results of 3 weeks, week 6 (W6), week 7 (W7), and week 8 (W8). The A-cells ([Figs. 4E] and [F]) and the C-cells ([Figs. 3D] and [E]) of the MNECs showed a very active secretory cell cycle in W6, W7 and W8. Both types exhibited a very strong staining affinity for AF and CHP. The oogonia remained normal, showed active proliferation, which was correlated with C-cells activity. At the end of W6, the abnormal changes in oocytes and trophocytes not only stopped increasing, but rather slightly decreased. Some abnormalities, such as degeneration, withdrawal of ooplasm, irregularities of plasma membranes of the oocytes, and disintegration in the trophocytes greatly decreased ([Fig. 4C]) in W7, and almost completely disappeared ([Fig. 4D]) in W8. The NSCs of the regenerated MNECs in these 3 weeks showed very strong activity and effectivity, both at the same time. The decrease and disappearance of the abnormalities are undoubtedly obvious signs of positive hormonal effects, of successful hormonal diffusion and transport, and of fully reconstructed blood capillary beds in the area of the regenerated NSCs. These facts are clearly supported and confirmed by histological preparations of A-cells and of the A-cells area ([Figs. 4E] and [F]). The A-cells showed a very strong neurosecretory cell cycle, whereas the area of A-cells exhibited a very rich supply of blood capillaries. Thus, the active A-cells supported by fully reconstructed blood capillary networks were able to show strong effectivity. These blood capillaries facilitated and ensured the diffusion and transport of A-cells neurosecretions from the MNECs to the oocytes and trophocytes in the ovaries, thereby triggering and restoring the vitellogenic and repairing processes. The repairing processes approached the peak at the end of W8, as it is clearly exhibited by the almost normal appearance of the ovarian tissue ([Fig. 4D]). These blood networks also supply the NSCs with oxygen, nutrients and, probably, hormones (stimulating and/or inhibiting), and finally remove the excretions and waste products from this area. These results are closely related to those reported in A.terrestris.[2] It was also emphasized that active NSCs were not able to exert positive hormonal effects without fully reconstructed blood capillary plexuses in the area of these cells. Therefore, a fully reconstructed blood capillary bed in the area of active regenerated NSCs is indispensable for successful hormonal diffusion, transport and effectivity.
Conclusions
The principal conclusions, which were deduced from the results of the present comprehensive experimental study, are: 1. Neurosecretion of A-cells of the SPPNG and not C-cells of the SBPNG and of the SNG controls the oocytes and trophocytes, based on several evidence: I. Several prominent abnormal changes appeared in oocytes and trophocytes, when A-cells were removed. II. The abnormal changes stopped increasing, started decreasing, underwent repair, and mostly disappeared, as soon as A-cells neurosecretion was transported to the ovaries (W6, W7, W8) via a fully reconstructed blood capillary plexus in the A-cells area. III. Abnormal changes continued increasing when the regeneration of fully-formed A-cells was delayed (W1, W2), or when the transport of A-cells neurohormone was delayed due to the lack (W3, W4) or scarcity (W5) of blood capillaries. IV. Presence of correlation between the strong neurosecretory activity of A-cells in both control and experimental worms and the active vitellogenic processes in oocytes and trophocytes. V. Neurosecretion of C-cells did not stop or decrease the abnormalities in oocytes and trophocytes. Therefore, C-cells play no role in vitellogenic or repairing processes. 2. Neurosecretion of the C-ells of the SBPNG and of the SNG, and of the A-cells of the SPPNG controls the oogonia due to three reasons: I. Oogonia were not affected at all by the removal and/or regeneration of A-cells. II. In W1 and W2, no active NSCs were present in experimental worms, except C-cells of the SNG, which remained very active throughout the tenure of the experiment and so did exactly the oogonia. III. Presence of a distinct contemporary positive correlation between the neurosecretory activity of C-cells and the active mitotic divisions of oogonia in both experimental and control worms. 3. Since the B-cells and U-cells of Hubl remained inactive throughout the tenure of the experiment and in both control and experimental worms, therefore, they have no role in oogonia proliferations, in vitellogenesis, and in the appearance or the disappearance of abnormalities. 4. The delay in hormonal effects of active A-cells was not due to low hormonal concentration or titer, since the number and activity of these cells increased exceedingly in W4 and W5, but even then they remained ineffective. Thus, the delay was undoubtedly due to the lack (W3, W4) and scarcity (W5) of blood capillaries in the A-cells regenerated area. Therefore, fully reconstructed blood capillary plexus in the regenerated area of NSCs is indispensable for hormonal diffusion, transport and effectivity. 5. Suprapharyngeal nerve ganglia are indispensable for reproduction, as they contain a huge number of A-cells, which elaborate a neurosecretion that controls normal vitellogenesis and leads to egg production. In absence of this gonadotropin, the oocytes and trophocytes will be destroyed, hence no eggs will be produced. 6. Removal of the MNECs in earthworms does not mean at all that all NSCs have been removed, since B-cells and C-cells are still present after the removal of the SPPNG in almost all SNG of the VNC due to redundancy.
No conflict of interest has been declared by the author(s).
Acknowledgments
Special thanks go to Miss Vivian Najem Shlemoon (M. Sc. IT, University of Wollongong, Australia), to Meelad Najem Shlemoon (MBBS, University of Mosul, now in Wollongong), and to Eugene Najem Shlemoon (Bachelor of Physical Education, University of Mosul) for their technical assistance. The authors also wish to acknowledge the support, help and encouragement of Dr. Wijdan M. S. Mero, Head of the Department of Biology, Dr. Maher Kh. Ali, Dean of the Faculty of Science, and Dr. Lazgin A. Jamil, President of the University of Zakho.
-
References
- 1 Durchon M. Recherches experimentale sur deux aspects de la reproduction chez les annelids. Polychetes: l'epitoque et la stolonisation. Annales des sciences naturelles Zoologie et Biologie Animale, XI 1952; 14: 117-206
- 2 Durchon M. L'endocrinologie des Vers et des Mollusques. Masson, Paris: 1967
- 3 Clark RB. Endocrinology and the reproductive biology of the Polychaetes. Oceanogr Mar Biol Annu Rev 1965; 3: 211-255
- 4 Hagadorn IR. Neurosecretion in the Hirudinea and its possible role in reproduction. Am Zool 1966; 6 (02) 251-261
- 5 Olive PJW, Bentley M. Hormonal control of oogenesis, ovulation and spawning in the annual reproductive cycle of the polychaete, Nephtys hombergii (Savigny, 1818) (Nephtyidae). International journal of Invertebrate Reproduction 1980; 2 (04) 205-221
- 6 Kulkarni GK, Anand CSK. Functional significance of supraesophageal and subesophageal ganglia in oogenesis of the freshwater leech, Hirudo birmanica (Blanchard, 1894). Comp Biochem Physiol Part A Physiol 1992; 103 (03) 545-550
- 7 Parthasarathi K, Ranganathan LS. Influence of pressmud on the development of the ovary, oogenesis and neurosecretory cells of the earthworm, Eudilus eugeniae (Kinberg). Afr Zool 2000; 35 (02) 281-286
- 8 Siekierska E. The structure of the ovary and oogenesis in gonadotropin-deprived earthworm, Dendrobaena veneta (Rosa). Zool Pol 2002; 47 (3–4): 37-48
- 9 Khalid VA. Seasonal changes in the histology of the ovary and its relation with neurosecretory activity in the earthworm, Dendrobaena atheca Cernosvitov. M. Sc. Thesis, Department of Biology, Faculty of Science, University of Zakho, Zakho, Kurdistan Regional Government, Iraq. 2014
- 10 Kiret ZT. The effects of the removal of the principal neurosecretory centers on the ovaries of the earthworm, Dendrobaena atheca Cernosvitov, (Annelida: Oligochaeta: Lumbricidae). M. Sc. Thesis, Department of Biology, Faculty of Science, University of Zakho, Zakho, Kurdistan Regional Government, Iraq. 2016
- 11 Gorgees NS. Study on the neurosecretory system of Dendrobaena atheca Cernosvitov and its role in regeneration. M. Sc. Thesis, Department of Biology, College of Science, University of Mosul, Mosul, Iraq. 1974
- 12 Baid IC, Gorgees NS. Presence of elementary neurohaemal organs in the lumbricid worm, Dendrobaena atheca Cernosvitov. J Anim Morphol Physiol 1975; 22 (02) 129-133
- 13 Baid IC, Gorgees NS. On the neurosecretory system of Dendrobaena atheca Cernosvitov. J Morphol 1977; 153 (02) 163-185
- 14 Gorgees NS, Baid IC. Presence of neurosecretory cells in the vegetative nervous system of Dendrobaena atheca Cernosvitov. Experientia 1975; 31 (04) 430-431
- 15 Golding DW. The infracerebral gland in Nephtys--a possible neuroendocrine complex. Gen Comp Endocrinol 1970; 14 (01) 114-126
- 16 Swiatek P. Ovary cord structure and oogenesis in Hirudo medicinalis and Haemopis sanguisuga (Clitellata, Annelida): remarks on different ovaries organization in Hirudinea. Zoomorphology 2008; 127: 213-226
- 17 Swiatek P, Krok F, Bielecki A. Germline cysts are formed during oogenesis in Eropobdella octaculata (Annelida, Clitellata, Erpobdellidae). Invertebr Reprod Dev 2010; 54: 53-63
- 18 Chaudhri PS, Banik D. Cytology and histology of the cerebral neurosecretory systems in some tropical earthworm species. J Cytol Histol 2015; 6: 1-7
- 19 Gorgees NS, Khalid VA. Histomorphology of the ovaries of the earthworm, Dendrobaena atheca Cernosvitov (Annelida, Clitellata: Oligochaeta). Journal of Morphological Sciences 2017; 34 (03) 178-185
- 20 Luna LG. Mnanual of histologic staining methods of the armed forces institute of pathology. 3rd ed. McGraw-Hill; New York: 1968
- 21 Galigher AE, Kozloff EN. Essentials of practical microtechnique. Lea and Febiger; Philadelphia: 1971
- 22 Culling CFA, Alliston RT, Barr WT. Cellular pathology techniques. 4th ed. London: Butterworth and Co. Ltd.; 1985
- 23 Al-Hajj HA. . Light microscope techniques: theory and practice, Jordan book center, Aman, Jordan. 1998
Address for correspondence
-
References
- 1 Durchon M. Recherches experimentale sur deux aspects de la reproduction chez les annelids. Polychetes: l'epitoque et la stolonisation. Annales des sciences naturelles Zoologie et Biologie Animale, XI 1952; 14: 117-206
- 2 Durchon M. L'endocrinologie des Vers et des Mollusques. Masson, Paris: 1967
- 3 Clark RB. Endocrinology and the reproductive biology of the Polychaetes. Oceanogr Mar Biol Annu Rev 1965; 3: 211-255
- 4 Hagadorn IR. Neurosecretion in the Hirudinea and its possible role in reproduction. Am Zool 1966; 6 (02) 251-261
- 5 Olive PJW, Bentley M. Hormonal control of oogenesis, ovulation and spawning in the annual reproductive cycle of the polychaete, Nephtys hombergii (Savigny, 1818) (Nephtyidae). International journal of Invertebrate Reproduction 1980; 2 (04) 205-221
- 6 Kulkarni GK, Anand CSK. Functional significance of supraesophageal and subesophageal ganglia in oogenesis of the freshwater leech, Hirudo birmanica (Blanchard, 1894). Comp Biochem Physiol Part A Physiol 1992; 103 (03) 545-550
- 7 Parthasarathi K, Ranganathan LS. Influence of pressmud on the development of the ovary, oogenesis and neurosecretory cells of the earthworm, Eudilus eugeniae (Kinberg). Afr Zool 2000; 35 (02) 281-286
- 8 Siekierska E. The structure of the ovary and oogenesis in gonadotropin-deprived earthworm, Dendrobaena veneta (Rosa). Zool Pol 2002; 47 (3–4): 37-48
- 9 Khalid VA. Seasonal changes in the histology of the ovary and its relation with neurosecretory activity in the earthworm, Dendrobaena atheca Cernosvitov. M. Sc. Thesis, Department of Biology, Faculty of Science, University of Zakho, Zakho, Kurdistan Regional Government, Iraq. 2014
- 10 Kiret ZT. The effects of the removal of the principal neurosecretory centers on the ovaries of the earthworm, Dendrobaena atheca Cernosvitov, (Annelida: Oligochaeta: Lumbricidae). M. Sc. Thesis, Department of Biology, Faculty of Science, University of Zakho, Zakho, Kurdistan Regional Government, Iraq. 2016
- 11 Gorgees NS. Study on the neurosecretory system of Dendrobaena atheca Cernosvitov and its role in regeneration. M. Sc. Thesis, Department of Biology, College of Science, University of Mosul, Mosul, Iraq. 1974
- 12 Baid IC, Gorgees NS. Presence of elementary neurohaemal organs in the lumbricid worm, Dendrobaena atheca Cernosvitov. J Anim Morphol Physiol 1975; 22 (02) 129-133
- 13 Baid IC, Gorgees NS. On the neurosecretory system of Dendrobaena atheca Cernosvitov. J Morphol 1977; 153 (02) 163-185
- 14 Gorgees NS, Baid IC. Presence of neurosecretory cells in the vegetative nervous system of Dendrobaena atheca Cernosvitov. Experientia 1975; 31 (04) 430-431
- 15 Golding DW. The infracerebral gland in Nephtys--a possible neuroendocrine complex. Gen Comp Endocrinol 1970; 14 (01) 114-126
- 16 Swiatek P. Ovary cord structure and oogenesis in Hirudo medicinalis and Haemopis sanguisuga (Clitellata, Annelida): remarks on different ovaries organization in Hirudinea. Zoomorphology 2008; 127: 213-226
- 17 Swiatek P, Krok F, Bielecki A. Germline cysts are formed during oogenesis in Eropobdella octaculata (Annelida, Clitellata, Erpobdellidae). Invertebr Reprod Dev 2010; 54: 53-63
- 18 Chaudhri PS, Banik D. Cytology and histology of the cerebral neurosecretory systems in some tropical earthworm species. J Cytol Histol 2015; 6: 1-7
- 19 Gorgees NS, Khalid VA. Histomorphology of the ovaries of the earthworm, Dendrobaena atheca Cernosvitov (Annelida, Clitellata: Oligochaeta). Journal of Morphological Sciences 2017; 34 (03) 178-185
- 20 Luna LG. Mnanual of histologic staining methods of the armed forces institute of pathology. 3rd ed. McGraw-Hill; New York: 1968
- 21 Galigher AE, Kozloff EN. Essentials of practical microtechnique. Lea and Febiger; Philadelphia: 1971
- 22 Culling CFA, Alliston RT, Barr WT. Cellular pathology techniques. 4th ed. London: Butterworth and Co. Ltd.; 1985
- 23 Al-Hajj HA. . Light microscope techniques: theory and practice, Jordan book center, Aman, Jordan. 1998










