Subscribe to RSS

DOI: 10.1055/s-0043-1774296
Primary Retroperitoneal Masses: A Pictorial Essay
Authors
Funding None.
Abstract
Primary retroperitoneal masses include a diverse and uncommon group of lesions that arise within the retroperitoneal space, but do not originate from any retroperitoneal organs. The majority of the lesions are malignant and imaging plays a pivotal role in the detection, staging, and preoperative planning. The evaluation of primary retroperitoneal masses is often challenging owing to the unfamiliarity with the common imaging features of various diseases affecting it. This article describes the multidetector computed tomography appearance of some primary retroperitoneal masses.
Introduction
Primary retroperitoneal masses may be categorized as solid or cystic and range from benign to aggressive in behavior. Benign and malignant masses should be distinguished whenever possible to avoid unnecessary surgical procedures.
Ultrasonography (USG) is the imaging modality performed initially due to its wide availability, ease of performance, low cost, and absence of radiation exposure. It plays an important role in characterization of mass lesions present in the retroperitoneum based on its location, size, shape, extent, margins, solid or cystic nature, echotexture, presence of calcification, and vascularity of the lesion on color Doppler. Presence of any free fluid and associated abnormality in any solid organ or enlarged lymph nodes can also be detected. However, USG examination of the retroperitoneum may be technically limited because of excessive bowel gas or body habitus.
Multidetector computed tomography (CT) plays an important role in depicting the exact compartmental localization of the masses into five retroperitoneal spaces, which include the anterior pararenal space, posterior pararenal space, perirenal space, central vascular space, and iliopsoas space.[1] It is currently the imaging modality of choice for the evaluation of morphology, internal characterization, assessment of disease extent, and involvement of vessels including the adjacent and distant structures.[2] It allows excellent reconstructions in planes other than the original scanning plane. Isotropic multiplanar reconstructions of thin overlapping slices in the coronal and sagittal plane provide excellent delineation of the location and extent of retroperitoneal masses.
The appearance of retroperitoneal lesions on cross-sectional imaging may pose a diagnostic challenge to the radiologist. Awareness of specific components of masses, tumor vascularity, and specific patterns of spread helps in further narrowing the differential diagnosis. Therefore, having knowledge of radiological features of various masses on cross-sectional imaging offers valuable insights for differential diagnosis of retroperitoneal masses and their management.
Normal Anatomy of Retroperitoneum
The retroperitoneum extends from the diaphragm to the pelvis. It extends between the posterior parietal peritoneum anteriorly and the fascia transversalis posteriorly.[2] The retroperitoneum is divided into the anterior pararenal space, posterior pararenal space, perirenal space, central vascular space, and iliopsoas space[1] ([Fig. 1]).


The anterior pararenal space contains the pancreas, duodenum, and a part of the ascending and descending colons. Kidneys, adrenal glands, blood vessels, lymphatics, and ureters are in perirenal spaces. Fat is only present in the posterior pararenal spaces. The central vascular compartment is located between the two perirenal spaces, behind the anterior pararenal space and in front of the spine. It extends from D12 to the L4–L5 vertebra and contains the inferior vena cava (IVC) and its afferent vasculature, abdominal aorta and its branches, lymphatic chains, and the abdominal sympathetic trunk. The iliopsoas compartment contains the psoas major, iliacus, and psoas minor muscles. Although the iliopsoas compartment is behind the transversalis fascia, it is still considered retroperitoneal because it is frequently involved in processes that begin in the retroperitoneum.[1]
The anterior and posterior pararenal spaces merge inferiorly, forming the infrarenal retroperitoneal space, which communicates with the prevesical space and extraperitoneal compartments of the pelvis. Because of loose connective tissue in the retroperitoneum, tumors can have widespread extension before clinical presentation.[3]
Solid primary retroperitoneal masses are broadly classified according to the tissue of origin. Four broad categories include mesodermal, neurogenic, germ cell, and lymphoid masses ([Table 1]).[3]
Cystic primary retroperitoneal masses are classified into neoplastic masses and non-neoplastic masses ([Table 2]).[3]
Malignant Retroperitoneal Tumors
In this section, we will review the imaging features of some common and uncommon malignant retroperitoneal masses such as malignant lymphoma, liposarcoma, myxofibrosarcoma (MFS), extra-adrenal neuroblastoma (NBL), and extra-skeletal Ewing's sarcoma.
Lymphoma
Lymphoma is the most common malignant retroperitoneal neoplasm, as well as being the most common small round cell tumor,[4] and typically presents as infiltrative homogenous hypovascular masses around the aorta or IVC, extending between and encasing structures without compressing them.[5] Upliftment of the great vessels results in “floating aorta” or “CT angiogram” sign ([Fig. 2]). Abdominal lymphomas are classified into solitary mass type, multiple nodular type, and diffuse type.[6] 18F-fluorodeoxyglucose (18F-FDG) positron emission tomography (PET)/CT is the standard examination for the diagnosis, staging, and in-treatment evaluation of lymphoma.[4] Heterogeneous or rim enhancement of lymph nodes due to necrosis with calcifications is seen following radiochemotherapy.[7] The main differential diagnosis for lymphomatous lymph nodes is tuberculosis.[6] Mesenteric lymph nodes are more commonly involved in tuberculosis. It is often associated with tuberculous peritonitis, which is characterized by omental caking, smudged mesentery, and high-density ascites. Mesenteric lymph nodes are not commonly involved in Hodgkin's disease. CT shows homogenous enhancement of the enlarged lymph nodes with fewer calcifications in untreated lymphoma cases. Peritoneal and omental lymphomatosis is not often seen.[6]


Liposarcoma
Liposarcomas are mesenchymal tumors arising from adipose tissue. Retroperitoneal liposarcomas are classified into five groups: well-differentiated liposarcoma, myxoid liposarcoma, round cell liposarcoma, pleomorphic liposarcoma, and dedifferentiated liposarcoma.[8]
Well-differentiated liposarcomas appear as well-defined fat containing masses with thin septa ([Fig. 3]). Calcifications or ossifications within a liposarcoma have proved to be a sign of poor prognosis, often indicating dedifferentiation.[9] Well-differentiated liposarcomas almost always undergo dedifferentiation,[8] which is suggested by the additional presence of a focal, nodular nonlipomatous region greater than 1 cm in size ([Fig. 4]).[10] Calcification is an important sign of dedifferentiation.[3] Myxoid liposarcomas appear as hypoechoic masses on USG. On CT and magnetic resonance imaging (MRI), myxoid liposarcomas have a “pseudocystic” appearance[3] due to the extracellular myxoid matrix. They appear as low-attenuation masses in comparison to muscle on CT with bright T2 signal on corresponding MRI. Slowly progressive reticular type of contrast enhancement is seen following contrast administration[3] ([Fig. 5]), a feature that distinguishes these lesions from a cystic mass.[10] Round cell liposarcoma and pleomorphic liposarcoma exhibit soft-tissue tumor attenuation and signal intensity with a minimal amount of fat.[8]






Myxofibrosarcoma
MFS are fibroblastic lesions that show a spectrum of cellularity, mitotic activity, and nuclear pleomorphism ranging from hypocellular lesions with minimal cytological atypia to more cellular lesions.[11] Low-grade MFS invariably show more than 75% myxoid change.[12] They have the unusual characteristic of extending along the fascial planes well beyond the primary center of the lesion.[13] Tail sign, which is defined by the tendency of the tumor to spread along the fascial planes in a curvilinear fashion, seen on MRI is a moderately sensitive and specific sign for MFS.[12] Tail sign is not only valuable for suggesting the diagnosis of MFS but its recognition is also essential in preoperative planning for complete tumor resection ([Fig. 6]).

Extra-Adrenal Neuroblastoma
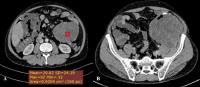
NBL, ganglioneuroblastoma, and ganglioneuroma are tumors of ganglion cell origin derived from the primordial neural crest cells.[14] The most common primary site for NBL is adrenal medulla (35%) and the extra-adrenal paraspinal ganglia (30–35%).[15] NBLs are heterogenous poorly circumscribed masses. Eighty percent to 90% show coarse, amorphous, and mottled calcifications ([Fig. 7]).[16] They can demonstrate extension across the midline and into adjacent body cavities. Vascular invasion is not a feature of NBLs.[17]


Extraskeletal Ewing's Sarcoma
Extraskeletal Ewing's sarcoma (EES) can occur anywhere in the body and can be classified into central/trunk and peripheral/extremity. On imaging, EESs show extensive necrosis and hemorrhage. Calcification is uncommon in EES. Tumors in the abdomen tend to have infiltrative margins with invasion of the adjacent structures and displace adjacent structures rather than encase them ([Fig. 8]). Retroperitoneal EESs are difficult to differentiate from other retroperitoneal tumors in adults such as the renal cell carcinoma and adrenocortical carcinoma. Features in favor of EESs are their earlier age of presentation, absence of metastatic lymphadenopathy, and calcification. Abdominal EESs are mostly unilateral and do not cross the midline.[18]


Benign Retroperitoneal Tumors
In this section, we will review the imaging features of some benign retroperitoneal masses like teratoma, paraganglioma, schwannoma, and urinoma.
Teratoma
Teratomas may be composed of mature or immature tissues deriving from the three pluripotent germ cell layers.[19] Mature cystic teratomas (MCTs) are characterized as cystic tumors with fat attenuation and Rokitansky's protuberance.[20] Rokitansky's nodules are soft-tissue protuberance within the cyst cavity. Bone or teeth, if present, tend to be located with Rokitansky's nodule ([Fig. 9]).[21] Immature teratomas (ITs) contain variable quantities of immature neural tissue. On cross-sectional imaging, ITs appear as large solid masses containing coarse calcifications and small foci of fat with concomitant hemorrhage.[22] Chemotherapy treatment of ITs can result in tissue maturation. This phenomenon is called “retroconversion”; the ITs look more like MCTs.[23] CT findings suggesting maturation are increasing density of tumor masses, appearance of some cystic components, fatty areas, and curvilinear and punctate calcifications. These masses do not invade the surrounding soft tissues or bone but may gradually compress them.[24]


Paraganglioma
Paragangliomas are rare tumors of chromaffin cell origin, arising from the extra-adrenal paraganglion cells of sympathetic or parasympathetic nervous systems. The most common site of paragangliomas in the abdomen is the organ of Zuckerkandl located in the para-aortic region near the origin of the inferior mesenteric artery ([Fig. 10]).[25] They appear as heterogeneous masses with necrosis, hemorrhage, and/or calcifications. Avid contrast material enhancement is often noted because of their hypervascular nature, especially peripheral.[26] On T2-weighted MRI, paragangliomas show diffuse high signal intensity known as “lightbulb” sign.


Schwannoma
Schwannomas are benign nerve sheath tumors of Schwann cell origin. On cross-sectional imaging, schwannomas are well-circumscribed masses, most commonly located in the paravertebral or presacral location. Target sign is seen on MRI, consisting of a central low to intermediate T2-signal intensity due to fibrous tissue surrounded by a peripheral high signal intensity of myxoid tissue.[27] It may be seen in both schwannomas and neurofibromas. Larger schwannomas are more likely to undergo degenerative changes, which include cyst formation, calcification, hemorrhage, and hyalinization ([Fig. 11]).[25] Ancient schwannoma refers to a long-standing lesion with advanced degenerative changes.[3]


Urinoma
Urinoma is a collection of extravasated urine that is found secondary to trauma or iatrogenic causes. They appear as well-defined lesions of fluid attenuation in the retroperitoneum, more commonly in the perirenal space. On CT, they are seen as well-circumscribed fluid collections showing increasing attenuation caused by contrast-enhanced urine entering the urinoma on excretory phases ([Fig. 12]).[25]


Conclusions
Primary retroperitoneal masses are a rare group of neoplasms. They pose a diagnostic dilemma on imaging due to their varied and overlapping appearances. Imaging features in combination with demographic and clinical information help in narrowing down the differential diagnosis. CT has the advantage of being easily available and has a short acquisition time. CT is valuable in characterizing these lesions, in evaluating their extent and relations with surrounding structures, which is useful for surgical planning.
Conflict of Interest
None declared.
-
References
- 1 Coffin A, Boulay-Coletta I, Sebbag-Sfez D, Zins M. Radioanatomy of the retroperitoneal space. Diagn Interv Imaging 2015; 96 (02) 171-186
- 2 Chinwan D, Vohra P. Role of multidetector computed tomography in evaluation of retroperitoneal masses. Int J Res Med Sci 2018; 6 (12) 3949-3953
- 3 Rajiah P, Sinha R, Cuevas C, Dubinsky TJ, Bush Jr WH, Kolokythas O. Imaging of uncommon retroperitoneal masses. Radiographics 2011; 31 (04) 949-976
- 4 Mota MMDS, Bezerra ROF, Garcia MRT. Practical approach to primary retroperitoneal masses in adults. Radiol Bras 2018; 51 (06) 391-400
- 5 Scali EP, Chandler TM, Heffernan EJ, Coyle J, Harris AC, Chang SD. Primary retroperitoneal masses: what is the differential diagnosis?. Abdom Imaging 2015; 40 (06) 1887-1903
- 6 Yu RS, Zhang WM, Liu YQ. CT diagnosis of 52 patients with lymphoma in abdominal lymph nodes. World J Gastroenterol 2006; 12 (48) 7869-7873
- 7 Manzella A, Borba-Filho P, D'Ippolito G, Farias M. Abdominal manifestations of lymphoma: spectrum of imaging features. ISRN Radiol 2013; 2013: 483069
- 8 Shaaban AM, Rezvani M, Tubay M, Elsayes KM, Woodward PJ, Menias CO. Fat-containing retroperitoneal lesions: imaging characteristics, localization, and differential diagnosis. Radiographics 2016; 36 (03) 710-734
- 9 Craig WD, Fanburg-Smith JC, Henry LR, Guerrero R, Barton JH. Fat-containing lesions of the retroperitoneum: radiologic-pathologic correlation. Radiographics 2009; 29 (01) 261-290
- 10 Murphey MD, Arcara LK, Fanburg-Smith J. From the archives of the AFIP: imaging of musculoskeletal liposarcoma with radiologic-pathologic correlation. Radiographics 2005; 25 (05) 1371-1395
- 11 Angervall L, Kindblom LG, Merck C. Myxofibrosarcoma. A study of 30 cases. Acta Pathol Microbiol Scand [A] 1977; 85A (02) 127-140
- 12 Lefkowitz RA, Landa J, Hwang S. et al. Myxofibrosarcoma: prevalence and diagnostic value of the “tail sign” on magnetic resonance imaging. Skeletal Radiol 2013; 42 (06) 809-818
- 13 Waters B, Panicek DM, Lefkowitz RA. et al. Low-grade myxofibrosarcoma: CT and MRI patterns in recurrent disease. AJR Am J Roentgenol 2007; 188 (02) W193-8
- 14 Rha SE, Byun JY, Jung SE, Chun HJ, Lee HG, Lee JM. Neurogenic tumors in the abdomen: tumor types and imaging characteristics. Radiographics 2003; 23 (01) 29-43
- 15 Papaioannou G, McHugh K. Neuroblastoma in childhood: review and radiological findings. Cancer Imaging 2005; 5 (01) 116-127
- 16 Xu Y, Wang J, Peng Y, Zeng J. CT characteristics of primary retroperitoneal neoplasms in children. Eur J Radiol 2010; 75 (03) 321-328
- 17 Dumba M, Jawad N, McHugh K. Neuroblastoma and nephroblastoma: a radiological review. Cancer Imaging 2015; 15 (01) 5
- 18 Somarouthu BS, Shinagare AB, Rosenthal MH. et al. Multimodality imaging features, metastatic pattern and clinical outcome in adult extraskeletal Ewing sarcoma: experience in 26 patients. Br J Radiol 2014; 87 (1038) 20140123
- 19 Outwater EK, Siegelman ES, Hunt JL. Ovarian teratomas: tumor types and imaging characteristics. Radiographics 2001; 21 (02) 475-490
- 20 Guinet C, Buy JN, Ghossain MA. et al. Fat suppression techniques in MR imaging of mature ovarian teratomas: comparison with CT. Eur J Radiol 1993; 17 (02) 117-121
- 21 Friedman AC, Pyatt RS, Hartman DS, Downey Jr EF, Olson WB. CT of benign cystic teratomas. AJR Am J Roentgenol 1982; 138 (04) 659-665
- 22 Bazot M, Cortez A, Sananes S, Boudghène F, Uzan S, Bigot J-M. Imaging of dermoid cysts with foci of immature tissue. J Comput Assist Tomogr 1999; 23 (05) 703-706
- 23 Aronowitz J, Estrada R, Lynch R, Kaplan AL. Retroconversion of malignant immature teratomas of the ovary after chemotherapy. Gynecol Oncol 1983; 16 (03) 414-421
- 24 Moskovic E, Jobling T, Fisher C, Wiltshaw E, Parsons C. Retroconversion of immature teratoma of the ovary: CT appearances. Clin Radiol 1991; 43 (06) 402-408
- 25 Goenka AH, Shah SN, Remer EM. Imaging of the retroperitoneum. Radiol Clin North Am 2012; 50 (02) 333-355 , vii
- 26 Al-Dasuqi K, Irshaid L, Mathur M. Radiologic-pathologic correlation of primary retroperitoneal neoplasms. Radiographics 2020; 40 (06) 1631-1657
- 27 Loke TK, Yuen NW, Lo KK, Lo J, Chan JC. Retroperitoneal ancient schwannoma: review of clinico-radiological features. Australas Radiol 1998; 42 (02) 136-138
Address for correspondence
Publication History
Article published online:
31 August 2023
© 2023. The Author(s). This is an open access article published by Thieme under the terms of the Creative Commons Attribution License, permitting unrestricted use, distribution, and reproduction so long as the original work is properly cited. (https://creativecommons.org/licenses/by/4.0/)
Thieme Medical and Scientific Publishers Pvt. Ltd.
A-12, 2nd Floor, Sector 2, Noida-201301 UP, India
-
References
- 1 Coffin A, Boulay-Coletta I, Sebbag-Sfez D, Zins M. Radioanatomy of the retroperitoneal space. Diagn Interv Imaging 2015; 96 (02) 171-186
- 2 Chinwan D, Vohra P. Role of multidetector computed tomography in evaluation of retroperitoneal masses. Int J Res Med Sci 2018; 6 (12) 3949-3953
- 3 Rajiah P, Sinha R, Cuevas C, Dubinsky TJ, Bush Jr WH, Kolokythas O. Imaging of uncommon retroperitoneal masses. Radiographics 2011; 31 (04) 949-976
- 4 Mota MMDS, Bezerra ROF, Garcia MRT. Practical approach to primary retroperitoneal masses in adults. Radiol Bras 2018; 51 (06) 391-400
- 5 Scali EP, Chandler TM, Heffernan EJ, Coyle J, Harris AC, Chang SD. Primary retroperitoneal masses: what is the differential diagnosis?. Abdom Imaging 2015; 40 (06) 1887-1903
- 6 Yu RS, Zhang WM, Liu YQ. CT diagnosis of 52 patients with lymphoma in abdominal lymph nodes. World J Gastroenterol 2006; 12 (48) 7869-7873
- 7 Manzella A, Borba-Filho P, D'Ippolito G, Farias M. Abdominal manifestations of lymphoma: spectrum of imaging features. ISRN Radiol 2013; 2013: 483069
- 8 Shaaban AM, Rezvani M, Tubay M, Elsayes KM, Woodward PJ, Menias CO. Fat-containing retroperitoneal lesions: imaging characteristics, localization, and differential diagnosis. Radiographics 2016; 36 (03) 710-734
- 9 Craig WD, Fanburg-Smith JC, Henry LR, Guerrero R, Barton JH. Fat-containing lesions of the retroperitoneum: radiologic-pathologic correlation. Radiographics 2009; 29 (01) 261-290
- 10 Murphey MD, Arcara LK, Fanburg-Smith J. From the archives of the AFIP: imaging of musculoskeletal liposarcoma with radiologic-pathologic correlation. Radiographics 2005; 25 (05) 1371-1395
- 11 Angervall L, Kindblom LG, Merck C. Myxofibrosarcoma. A study of 30 cases. Acta Pathol Microbiol Scand [A] 1977; 85A (02) 127-140
- 12 Lefkowitz RA, Landa J, Hwang S. et al. Myxofibrosarcoma: prevalence and diagnostic value of the “tail sign” on magnetic resonance imaging. Skeletal Radiol 2013; 42 (06) 809-818
- 13 Waters B, Panicek DM, Lefkowitz RA. et al. Low-grade myxofibrosarcoma: CT and MRI patterns in recurrent disease. AJR Am J Roentgenol 2007; 188 (02) W193-8
- 14 Rha SE, Byun JY, Jung SE, Chun HJ, Lee HG, Lee JM. Neurogenic tumors in the abdomen: tumor types and imaging characteristics. Radiographics 2003; 23 (01) 29-43
- 15 Papaioannou G, McHugh K. Neuroblastoma in childhood: review and radiological findings. Cancer Imaging 2005; 5 (01) 116-127
- 16 Xu Y, Wang J, Peng Y, Zeng J. CT characteristics of primary retroperitoneal neoplasms in children. Eur J Radiol 2010; 75 (03) 321-328
- 17 Dumba M, Jawad N, McHugh K. Neuroblastoma and nephroblastoma: a radiological review. Cancer Imaging 2015; 15 (01) 5
- 18 Somarouthu BS, Shinagare AB, Rosenthal MH. et al. Multimodality imaging features, metastatic pattern and clinical outcome in adult extraskeletal Ewing sarcoma: experience in 26 patients. Br J Radiol 2014; 87 (1038) 20140123
- 19 Outwater EK, Siegelman ES, Hunt JL. Ovarian teratomas: tumor types and imaging characteristics. Radiographics 2001; 21 (02) 475-490
- 20 Guinet C, Buy JN, Ghossain MA. et al. Fat suppression techniques in MR imaging of mature ovarian teratomas: comparison with CT. Eur J Radiol 1993; 17 (02) 117-121
- 21 Friedman AC, Pyatt RS, Hartman DS, Downey Jr EF, Olson WB. CT of benign cystic teratomas. AJR Am J Roentgenol 1982; 138 (04) 659-665
- 22 Bazot M, Cortez A, Sananes S, Boudghène F, Uzan S, Bigot J-M. Imaging of dermoid cysts with foci of immature tissue. J Comput Assist Tomogr 1999; 23 (05) 703-706
- 23 Aronowitz J, Estrada R, Lynch R, Kaplan AL. Retroconversion of malignant immature teratomas of the ovary after chemotherapy. Gynecol Oncol 1983; 16 (03) 414-421
- 24 Moskovic E, Jobling T, Fisher C, Wiltshaw E, Parsons C. Retroconversion of immature teratoma of the ovary: CT appearances. Clin Radiol 1991; 43 (06) 402-408
- 25 Goenka AH, Shah SN, Remer EM. Imaging of the retroperitoneum. Radiol Clin North Am 2012; 50 (02) 333-355 , vii
- 26 Al-Dasuqi K, Irshaid L, Mathur M. Radiologic-pathologic correlation of primary retroperitoneal neoplasms. Radiographics 2020; 40 (06) 1631-1657
- 27 Loke TK, Yuen NW, Lo KK, Lo J, Chan JC. Retroperitoneal ancient schwannoma: review of clinico-radiological features. Australas Radiol 1998; 42 (02) 136-138




























