Subscribe to RSS
Please copy the URL and add it into your RSS Feed Reader.
https://www.thieme-connect.de/rss/thieme/en/10.1055-s-00000083.xml
Synlett 2014; 25(16): 2233-2238
DOI: 10.1055/s-0034-1378324
DOI: 10.1055/s-0034-1378324
synpacts
α-Amination of Phosphonates: A Direct Synthesis of α-Amino Phosphonic Acids and Their Derivatives
Authors
Further Information
Publication History
Received: 02 May 2014
Accepted after revision: 20 May 2014
Publication Date:
07 July 2014 (online)
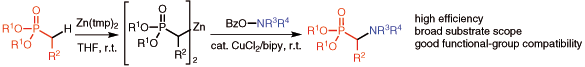
Abstract
A direct approach to access important α-amino phosphonic acids and their derivatives through the formation of C–N bonds is described. Several α-amination reactions of phosphonates that involve electrophilic aminating agents are reviewed. A recent development is highlighted in which an acyclic or cyclic amine is introduced into the α-position of a phosphonate in one step through deprotonative formation of an α-zincate followed by copper-catalyzed amination with an O-acylhydroxylamine.
-
References
- 1 Mucha A, Kafarski P, Berlicki Ł. J. Med. Chem. 2011; 54: 5955
- 2a Forlani G, Occhipinti A, Berlicki Ł, Dziędzioła G, Wieczorek A, Kafarski P. J. Agric. Food Chem. 2008; 56: 3193
- 2b Forlani G, Berlicki Ł, Duò M, Dziędzioła G, Giberti S, Bertazzini M, Kafarski P. J. Agric. Food Chem. 2013; 61: 6792
- 3a Chen M.-H, Chen Z, Song B.-A, Bhadury PS, Yang S, Cai X.-J, Hu D.-Y, Xue W, Zeng S. J. Agric. Food Chem. 2009; 57: 1383
- 3b Hu D.-Y, Wan Q.-Q, Yang S, Song B.-A, Bhadury PS, Jin L.-H, Yan K, Liu F, Chen Z, Xue W. J. Agric. Food Chem. 2008; 56: 998
- 3c Long N, Cai X.-J, Song B.-A, Yang S, Chen Z, Bhadury PS, Hu D.-Y, Jin L.-H, Xue W. J. Agric. Food Chem. 2008; 56: 5242
- 4a Allen JG, Atherton FR, Hall MJ, Hassall CH, Holmes SW, Lambert RW, Nisbet LJ, Ringrose PS. Nature 1978; 272: 56
- 4b Atherton FR, Hassall CH, Lambert RW. J. Med. Chem. 1986; 29: 29
- 5a Kafarski P, Lejczak B. Curr. Med. Chem.: Anti-Cancer Agents 2001; 1: 301
- 5b Lavielle G, Hautefaye P, Schaeffer C, Boutin JA, Cudennec CA, Pierre A. J. Med. Chem. 1991; 34: 1998
- 6a Allen MC, Fuhrer W, Tuck B, Wade R, Wood JM. J. Med. Chem. 1989; 32: 1652
- 6b Bird J, De Mello RC, Harper GP, Hunter DJ, Karran EH, Markwell RE, Miles-Williams AJ, Rahman SS, Ward RW. J. Med. Chem. 1994; 37: 158
- 6c Smith WW, Bartlett PA. J. Am. Chem. Soc. 1998; 120: 4622
- 7a Kafarski P, Lejczak B. Phosphorus, Sulfur Silicon Relat. Elem. 1991; 63: 193
- 7b Kudzin ZH, Kudzin MH, Drabowicz J, Stevens CV. Curr. Org. Chem. 2011; 15: 2015
- 7c Kukhar VP, Soloshonok VA, Solodenko VA. Phosphorus, Sulfur Silicon Relat. Elem. 1994; 92: 239
- 8a Abell JP, Yamamoto H. J. Am. Chem. Soc. 2008; 130: 10521
- 8b Davis FA, Lee S, Yan HX, Titus DD. Org. Lett. 2001; 3: 1757
- 8c Ingle GK, Liang YX, Mormino MG, Li G, Fronczek FR, Antilla JC. Org. Lett. 2011; 13: 2054
- 8d Joly GD, Jacobsen EN. J. Am. Chem. Soc. 2004; 126: 4102
- 8e Lefebvre IM, Evans SA. Jr. J. Org. Chem. 1997; 62: 7532
- 8f Nakamura S, Hayashi M, Hiramatsu Y, Shibata N, Funahashi Y, Toru T. J. Am. Chem. Soc. 2009; 131: 18240
- 8g Pudovik AN. Dokl. Akad. Nauk SSSR 1952; 74: 1528
- 8h Pudovik AN, Konovalova I. Synthesis 1979; 81
- 8i Sasai H, Arai S, Tahara Y, Shibasaki M. J. Org. Chem. 1995; 60: 6656
- 8j Smith AB. III, Yager KM, Taylor CM. J. Am. Chem. Soc. 1995; 117: 10879
- 8k Yin L, Bao Y, Kumagai N, Shibasaki M. J. Am. Chem. Soc. 2013; 135: 10338
- 9 Gao Y, Huang Z, Zhuang R, Xu J, Zhang P, Tang G, Zhao Y. Org. Lett. 2013; 15: 4214
- 10a Fields EK. J. Am. Chem. Soc. 1952; 74: 1528
- 10b Kabachnik MI, Medved TY. Bull. Acad. Sci. USSR, Div. Chem. Sci. (Engl. Transl.) 1953; 2: 769
- 10c Cheng X, Goddard R, Buth G, List B. Angew. Chem. Int. Ed. 2008; 47: 5079
- 11a Groth U, Richter L, Schöllkopf U. Tetrahedron 1992; 48: 117
- 11b Hanessian S, Bennani YL. Tetrahedron Lett. 1990; 31: 6465
- 11c Hannour S, Roumestant ML, Viallefont P, Riche C, Martinez J, El Hallaoui A, Ouazzani F. Tetrahedron: Asymmetry 1998; 9: 2329
- 11d Kobayashi S, Kiyohara H, Nakamura Y, Matsubara R. J. Am. Chem. Soc. 2004; 126: 6558
- 12a Burk MJ, Stammers TA, Straub JA. Org. Lett. 1999; 1: 387
- 12b Goulioukina NS, Shergold IA, Bondarenko GN, Ilyin MM, Davankov VA, Beletskaya IP. Adv. Synth. Catal. 2012; 354: 2727
- 12c Gridnev ID, Yasutake M, Imamoto T, Beletskaya IP. Proc. Natl. Acad. Sci. U. S. A. 2004; 101: 5385
- 12d Hammerschmidt F, Hanbauer M. J. Org. Chem. 2000; 65: 6121
- 13a Erdik E. Tetrahedron 2004; 60: 8747
- 13b Ciganek E. Electrophilic Amination of Carbanions, Enolates, and Their Surrogates. In Organic Reactions. John Wiley & Sons, Inc; Hoboken, NJ: 2009
- 13c Denmark SE, Chatani N, Pansare SV. Tetrahedron 1992; 48: 2191
- 13d Jommi G, Miglierini G, Pagliarin R, Sello G, Sisti M. Tetrahedron 1992; 48: 7275
- 13e Hanessian S, Bennani YL. Synthesis 1994; 1272
- 13f Pagliarin R, Papeo G, Sello G, Sisti M, Paleari L. Tetrahedron 1996; 52: 13783
- 13g Jiang Q, Ren Y, Feng J. J. Neurosci. 2008; 28: 12993
- 13h Greck C, Drouillat B, Thomassigny C. Eur. J. Org. Chem. 2004; 1377
- 13i Bernardi L, Zhuang W, Jørgensen KA. J. Am. Chem. Soc. 2005; 127: 5772
- 14 McDonald SM, Wang Q. Angew. Chem. Int. Ed. 2014; 53: 1867
- 15a Hendrick CE, McDonald SL, Wang Q. Org. Lett. 2013; 15: 3444
- 15b Huang H.-T, Lacy TC, Błachut B, Ortiz GX. Jr, Wang Q. Org. Lett. 2013; 15: 1818
- 16 McDonald SL, Wang Q. Chem. Commun. 2014; 50: 2535
- 17a Hlavinka ML, Greco JF, Hagadorn JR. Chem. Commun. 2005; 5304
- 17b Hlavinka ML, Hagadorn JR. Organometallics 2007; 26: 4105
- 17c Rees WS. Jr, Just O, Schumann H, Weimann R. Polyhedron 1998; 17: 1001
- 18a Berman AM, Johnson JS. J. Am. Chem. Soc. 2004; 126: 5680
- 18b Berman AM, Johnson JS. Synlett 2005; 1799
- 18c Berman AM, Johnson JS. J. Org. Chem. 2006; 71: 219
- 18d Campbell MJ, Johnson JS. Org. Lett. 2007; 9: 1521
- 19 Matsda N, Hirano K, Satoh T, Miura M. Angew. Chem. Int. Ed. 2012; 51: 11827
- 20 Noack M, Göttlich R. Chem. Commun. 2002; 536

